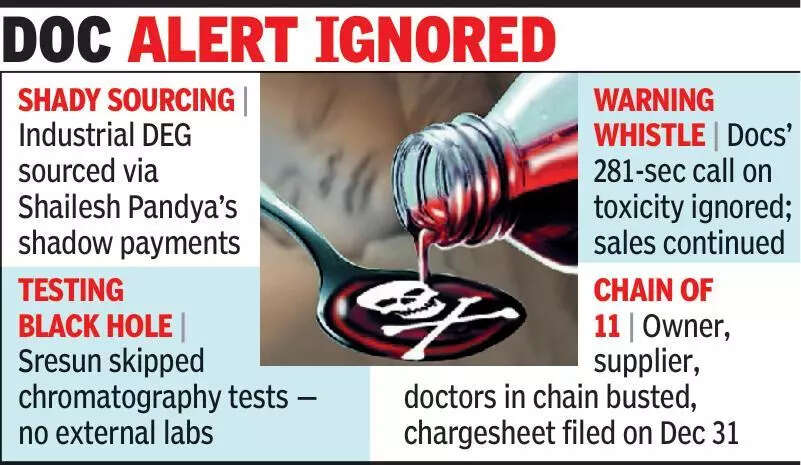
BHOPAL: Batch SR-13 – the ‘killer batch' of cough syrup Coldrif – that took the lives of over 25 children in MP's Chhindwara district in Oct last year, moved freely through the supply, manufacturing and distribution chain without being stopped, from procurement of raw material to retail sale, investigators have mentioned in the case chargesheet.
The chargesheet, a copy of which is with TOI, was filed on Dec 31, last year in the court of additional sessions judge in Parasia in MP's Chhindwara district.
Watch
Bhopal: Grisly Murder Case, Infamous Fugitive Captured, Unusual PCC Drama
The chargesheet described the events as a "continuous and interlinked chain" rather than an isolated lapse. From sourcing decisions and financial transactions to testing gaps and retail-level breakdowns, the chargesheet presented a sequence in which Batch SR-13 cleared every gate placed before it, until it was too late.
According to the chargesheet, the trail began with the procurement of raw material allegedly supplied by Shailesh Pandya, an accused in the case. He was later arrested. The document stated that industrial-grade material was sourced for use in the syrup manufactured by Sresun Pharma. Financial records examined during the probe, the chargesheet noted, reflected that payments were not made against these supplies in a transparent manner.
The payments were not made directly to the supplier but were routed through other channels.
Investigators pointed to the pricing pattern and documentation surrounding the transactions as indicative of awareness that the material was not standard pharmaceutical grade.
The chargesheet then recorded that the company did not have adequate and standard testing facilities, including gas chromatography capability required to detect contaminants such as Diethylene Glycol (DEG), a substance historically associated with toxicity in liquid formulations. Nor was there an accredited external laboratory arrangement to compensate for this gap, the chargesheet said.
Despite this, Batch SR-13 was cleared and released. Once dispatched, the batch entered the distribution network. The chargesheet detailed irregularities at the stockist and retail levels, including invoices that did not align with manufacturing timelines, missing bottles, absence of prescription-linked sale, and alleged destruction of leftover stock after concerns emerged.
The investigation recorded a critical moment at this stage. As unusual acute kidney injury cases began surfacing in Parasia, a 281-second phone call took place between a local doctor and an external medical expert. The chargesheet noted that during this conversation, the possibility of drug-induced toxicity, including reference to earlier DEG-linked tragedies, was discussed.
Yet, the document stated, no immediate regulatory escalation followed, and the syrup continued to remain in circulation within the same distribution network.