- News
- India News
- Serum institute submits data from trials of vaccine
Trending
This story is from December 23, 2020
Serum institute submits data from trials of vaccine
Sushmi Dey / TNN / Dec 23, 2020, 04:36 IST
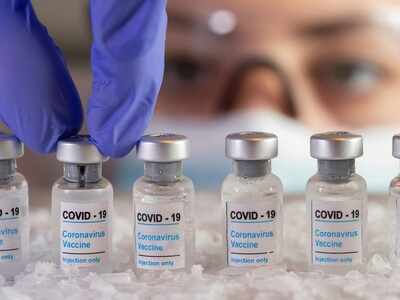
The Subject Expert Committee (SEC) on Covid vaccine under the Drugs Controller General of India had asked Bharat Biotech and SII to produce updated data from ongoing clinical trials in India.
The latest development may once again set the clock ticking for the launch of the vaccine, as the government is expecting to roll out vaccination against Covid-19 by mid-January.
End of Article
Follow Us On Social Media
Visual Stories
Photostories
Tired of too many ads?go ad free now
Trending Stories
In India
Entire Website
- Government to rank districts on road safety parameters
- Dust storm, heavy rain sweeps across Delhi-NCR, metal structure at Delhi airport collapses
- Pakistan PM, president pledge 'befitting response' to any act of aggression amid rising tensions with India after Pahalgam attack
- Congress dismisses caste census announcement as ‘diversionary tactic’ of government
- Government call on caste count game changer, exposes Congress: BJP
- SC rejects probe plea, says don’t file petitions to demoralise forces
- US clears $131 million sale of defence software, equipment to India
- Navy intensifies muscle-flexing against Pakistan even as small arms fire continues across LoC
- Terror response a bigger issue than caste census: Congress
- Tweak PMLA, make hawala operators & angadias partners in probe: Law officer
- Taylor Swift breaks silence on social media as Travis Kelce partied in Vegas with Jason Kelce and Justin Timberlake
- Skylar Diggins confirms removing her husband’s name as she embraces a new chapter with Seattle Storm
- Apple CEO on US-made iPhones: I don’t like to employ all these people foreign, but...
- Baba Vanga’s 2025 predictions: Is the next world war just around the corner - here’s what she predicted about the global wars and other crisis
- Kirk Cousins could end Aaron Rodgers' comeback dreams as Steelers move fast to replace their QB gap
- Amazon CEO Andy Jassy to business leaders in the US on Trump tariffs: At the end of the day, we have a …
- No, Michelle Obama didn’t call herself a black man; viral clip takes her words out of context
- Google CEO Sundar Pichai makes an ‘Apple announcement’ under oath, tells court: Tim Cook was trying to....
- Tyreek Hill’s wife Keeta Vaccaro shares “recovery days” post after filing for divorce and accusing the NFL star of “neglecting” their child
- Shedeur Sanders' net worth 2025: How much money does the new Browns quarterback have?
Tired of too many ads?go ad free now
Explore Every Corner
Across The Globe
Tyreek HillXi JinpingBoard Exam Result 2025Carmelo AnthonyCBSE Board Exam ResultsNEET UG Admit CardPalwasha Mohammad Zai KhanRR vs MI Live ScoreIPL Match TodayPahalgam Terror AttackShannon SharpeSatya NadellaKamala HarrisJung Hoo LeeKirk CousinsShedeur Sanders Net WorthMohsen MahdawiSambhavi JaiswalJalen BrunsonMartina SablikovaBank Holidays MayElon MuskOscar PiastriAntonio BrownShannon SharpeCharlotte FlairIPL Purple CapIPL Orange CapIPL Schedule 2025IPL Points Table
Hot on the Web
Hit 3 Movie ReviewRetro ReviewRaid 2 Movie ReviewRetro Box Office CollectionRetro OTT ReleaseRaid 2 Box Office CollectionShankaracharya Jayanti WishesHit 3 Movie ReviewHIT 3 Twitter ReviewRaid 2 Twitter ReviewRetro Twitter ReviewJaat Box Office CollectionMumtazToxic Cooking OilsKidney Damage HabitsSaif Ali KhanMark CarneyLow Maintenance SucculentsParesh RawalWorld Oldest Living AnimalsUnique Green Colour BirdsHarmful Supplements For KidneyRaid 2 Social Media ReviewMeghan MarkleBrain ExercisesChhaya KadamHair Growth OilsRaid 2 Movie ReviewKapil SharmaUS Do Not Travel AdvisorySharp Brain ExercisesHania AamirMeghan MarkleCancer May HoroscopeLeo May HoroscopeVirgo May HoroscopeLibra May HoroscopeScorpio May HoroscopeSagittarius May HoroscopeCapricorn May HoroscopeAquarius May HoroscopePisces May Horoscope
Trending Topics
Sundar PichaiRajnath SinghAsaduddin OwaisiMI vs RRHarbhajan SinghJEEP 2025 RegistrationKSEAB Karnataka SSLC Result 2025Sanju SamsonCaste CensusCBSE Board ResultsPakistan Air ForceBill Belichicks GirlfriendRohit SharmaVaibhav SuryavanshiShohei OhtaniWWE Backlash 2025 PredictionsSignal App Security ScandalTim CookSocial Security CardsPlaystation Plus Monthly GamesAnthony StolarzProject Egoist CodesOne Piece Episode 1128Wordle TodayNYT ConnectionsApple iPhone 16UPS LayoffsNyt StrandsElon MuskDilip GhoshTyrese HaliburtonVanessa BryantYuzvendra ChahalMohsen MahdawiJrue HolidayVirat KohliTaylor SwiftBiomass SatelliteWinnipeg JetsSuryakumar YadavPublic Holidays AprilAmazon Sale
Popular Categories
HeadlinesSports NewsBusiness NewsIndia NewsWorld NewsBollywood NewsHealth+ TipsIndian TV ShowsTechnologyTravelEtimesHealth & FitnessWWENFLPahalgam Terror AttackAstrologyWeather TodayIPLAnimeApril Bank HolidaysPublic Holidays in AprilIPL TeamsStock Market HolidaysNSE HolidaysBSE HolidaysMCX HolidaysTechnology NewsInternational SportsPublic HolidaysBank Holidays
Latest News
Luka Doncic’s Los Angeles Lakers Future in Doubt After Playoff Exit: Will He Stay or Go?What is the net worth of Mike Waltz?'142K illegal aliens vs Donald Trump': White House joins '100 men vs gorilla' viral meme trend to highlight US immigration recordGovernment to rank districts on road safety parametersWho is Kavan Markwood? Ex-football player identified as fan who fell 21 feet at Pirates-Cubs game; viral video stuns netizens'Ground Zero' Box Office Collection day 7: Emraan Hashmi’s film sees sharp DROP, earns just Rs 7 lakh as Ajay Devgn's 'Raid 2' hits cinemasJJ Redick indirectly blasts Luka Doncic’s ‘lack of fitness’ for Los Angeles Lakers’ playoff collapse? - "There are ones that could have been in better shape!"Who is Chandra Holt? Former Walmart exec in spotlight as Kohl’s CEO Ashley Buchanan fired over vendor dealNBA Trade Rumors: Los Angeles Lakers could acquire $135 million Boston Celtics superstar to form Big 3 with LeBron James and Luka Doncic and bolster title hopesDust storm, heavy rain sweeps across Delhi-NCR‘Raid 2’ Box Office collection day 1: Ajay Devgn’s crime thriller sequel earns Rs 18.25 crore, faces tough competition from ‘HIT 3’ and ‘Retro’What is going on in the Wind Breaker manga right now? Spoilers Ahead!!!Why a 10-minute walk after work is highly recommendedESPN's Kendrick Perkins calls Lakers’ Luka Doncic the "worst defensive star" in modern NBAJay Feely, former NFL kicker and current broadcaster wants Stephen A. Smith to run for PresidentHenry Ruggs training in prison as he plans an unlikely NFL comeback story, reveals Josh JacobsNBA HOFer Declares Los Angeles Lakers' Championship Hinges on Passing Torch from LeBron James to Luka Doncic After Loss to Timbs - "You have to get out of the constraints!"Bill Belichick’s relationship with 24-year-old Jordon Hudson reportedly brought the softer side and major personal changes in his life









